|
Now, using the information from the argon box we know that the atomic mass of argon is 39.948.Īfter rounding it up to the nearest whole number we get 40.Īlso, as calculated above, the number of protons in the argon atom is 18. Number of neutrons = Atomic mass (rounding it up to the nearest whole number) – Number of protons The formula for calculating the number of neutrons present in an atom is given below: Moving on, we will now calculate the number of neutrons in the argon atom. Therefore, for the argon atom, the number of protons = atomic number = 18 In the case of the argon atom, the atomic number is 18. The number of protons for any atom is always equal to the atomic number of that atom. Now, using the above information we will draw the Bohr atomic model for the argon atom.įor this, we will first have to calculate the number of atomic species. The electronic configuration of chlorine is 3s 23p 6.The information that we can derive from the above-mentioned Argon box is as follows: Argon AtomĪrgon is a noble gas located in group 18 of the periodic table: The electrons revolve around the nucleus in K, L, and M shells. The argon atom contains 22 neutrons, 18 protons, and 18 electrons. The electrons are also allowed to hop from lower to higher as they gain energy or fall from higher to lower energy levels as they lose their energy. These electrons are responsible for the formation of bonds. 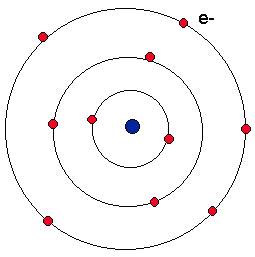
The electrons located in the outermost shell carry maximum energy and are also referred to as valence electrons. the shell closest to the nucleus are said to be in the ground state and carry minimum energy. 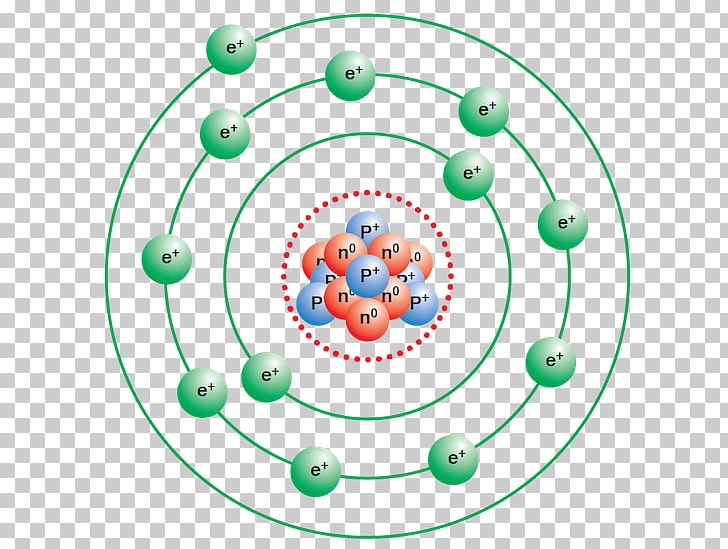
Therefore, the electrons located in the K-shell i.e. This is why the shells are also known as energy levels. The energy of the electrons also increases as the number of shells increases. This number increases away from the nucleus. In the Bohr model of the atom, the shells are named as K, L, M, N, etc., or 1, 2, 3, 4, etc. Only a fixed number of electrons are allowed to follow a particular orbit owing to the difference in their energy and capacity of the shell to accommodate the electrons. Shells: The path taken by the electrons around the nucleus are termed as shells or orbits.The location of an electron with respect to the nucleus depends upon the energy of the electron. Electrons: The negatively charged entities that move in fixed circular paths orbiting the nucleus of an atom.These are denoted by using the symbol n°. Neutrons: These are the neutral entities present inside the nucleus of an atom and are responsible for most physical properties of atoms.Protons: These are the positively charged entities present in the nucleus of an atom and are represented through the symbol p +.It derives its positive charge owing to the positively charged protons. Nucleus: The centre or core of the atom comprising neutrons and protons.electrons, protons, and neutrons.īefore we dig deep into the Bohr model of the atom we should first understand a few important terms related to this model used in describing the structure of an atom. 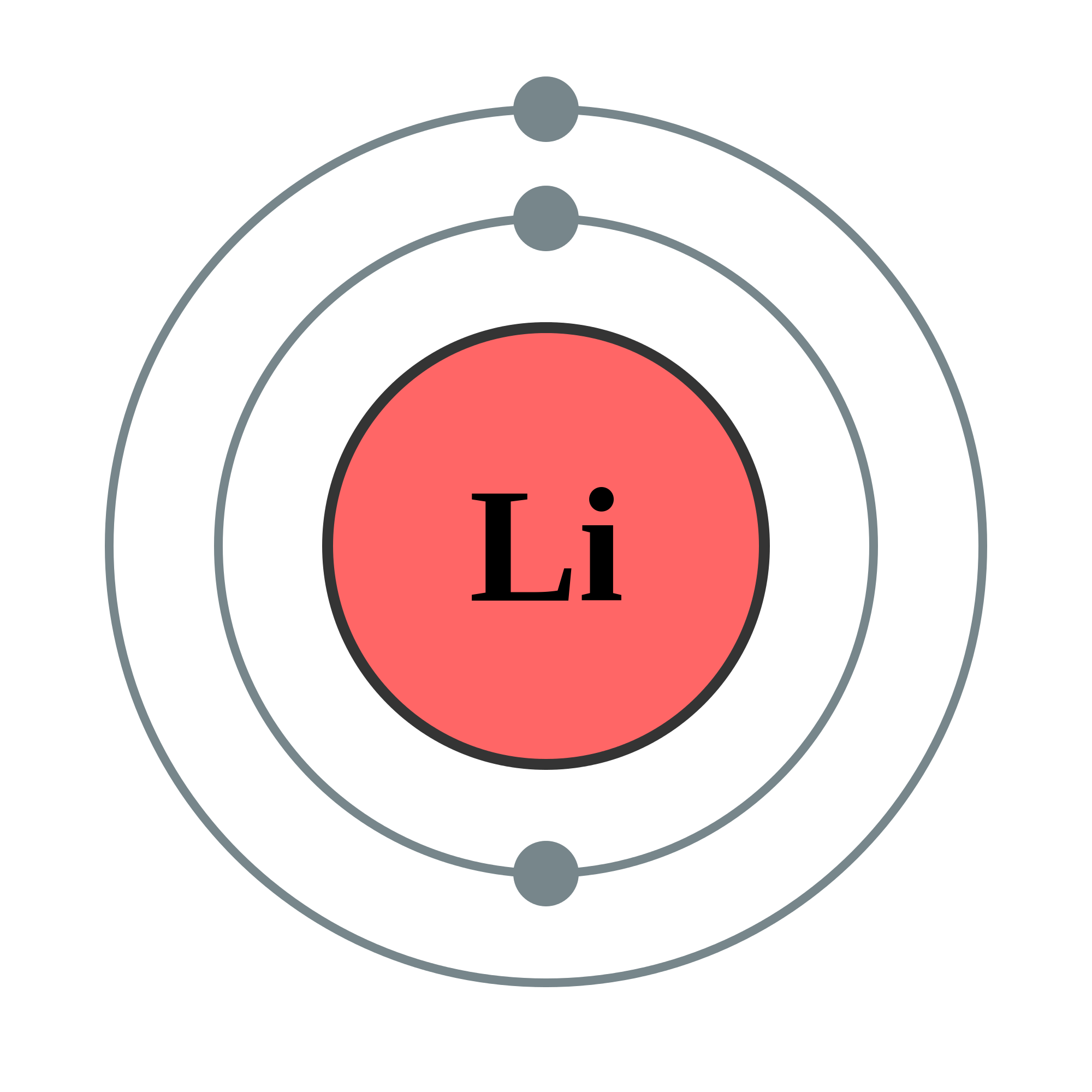
The Bohr model of atom defines the atomic structure of elements through a pictorial representation illustrating all the atomic particles viz. In this model, he postulated that every electron moves in a pre-decided orbit having fixed size and energy. Niel Bohr modified this model and presented the improvised Bohr-Rutherford model of the atom in 1913. The Rutherford model of the atom proposed in 1911 could not explain the stability of electrons while moving around the nucleus.Īs per the classical mechanics and electromagnetic theory such particles could not remain stable and would lose energy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
